The Diabetes Study Changed the Question
Why deterministic convergence matters more than another variant list
Most genomics still asks a reductionist question: which variant is associated with disease?
That question has been useful, but it is no longer sufficient. It is not sufficient for explaining why apparently strong signals disappear across cohorts. It is not sufficient for explaining why interventions built around a single target so often disappoint. And it is not sufficient for describing the behavior we observed in this diabetes study.
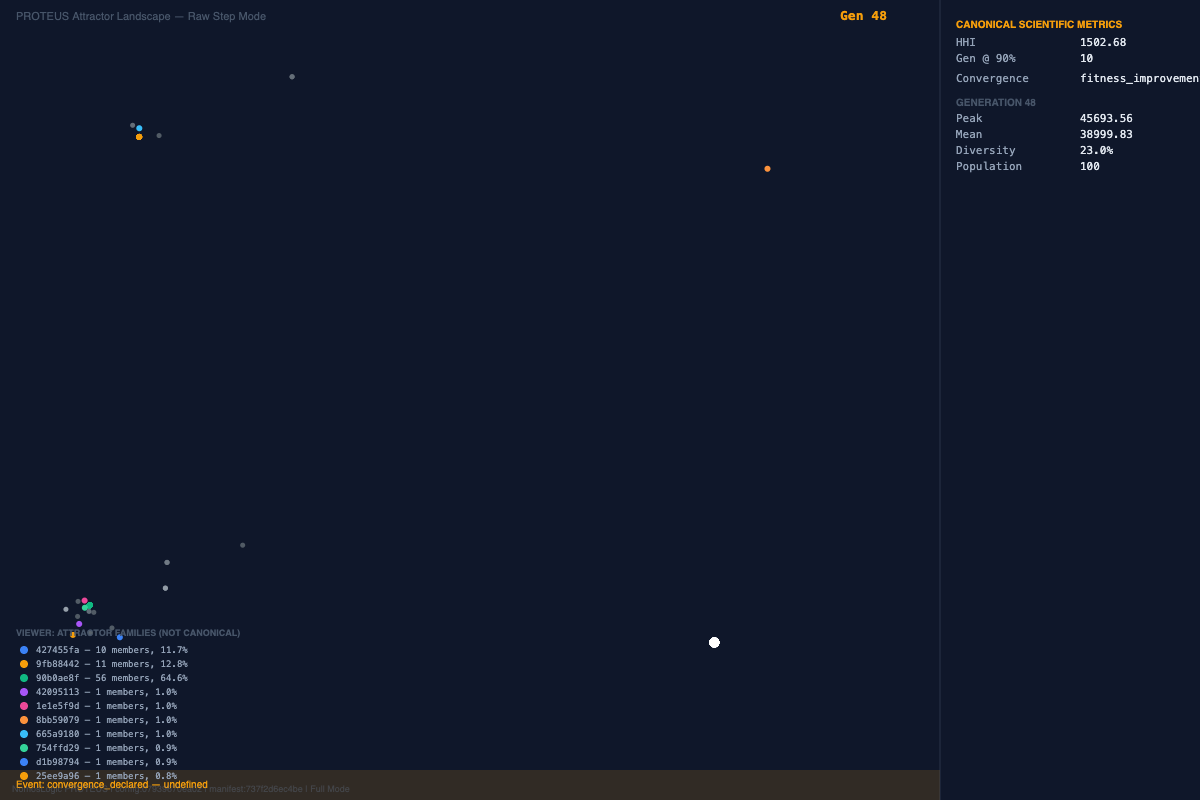
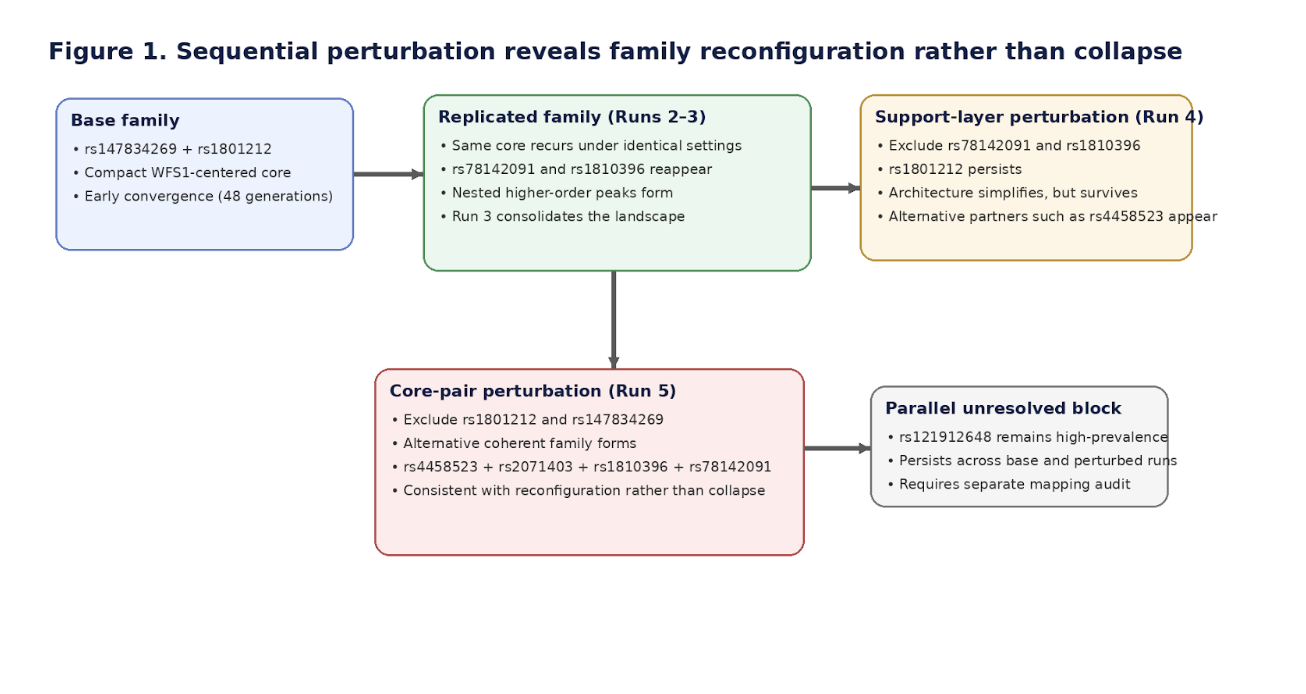
What Proteus surfaced was not simply a ranked list of loci. It surfaced repeated convergence on stable peak families under identical conditions, followed by orderly reconfiguration under targeted exclusion. In other words, the system behaved less like a brittle stack of isolated risk markers and more like a constrained architecture capable of preserving structure under pressure.
That distinction matters. If the architecture reorganizes rather than collapses, then the biological question changes. We are no longer asking which single variant explains the phenotype. We are asking which interacting structures are stable, which are replaceable, which are compensatory, and which disruptions actually break the system.
That is the real significance of this diabetes series.
What the diabetes runs actually showed
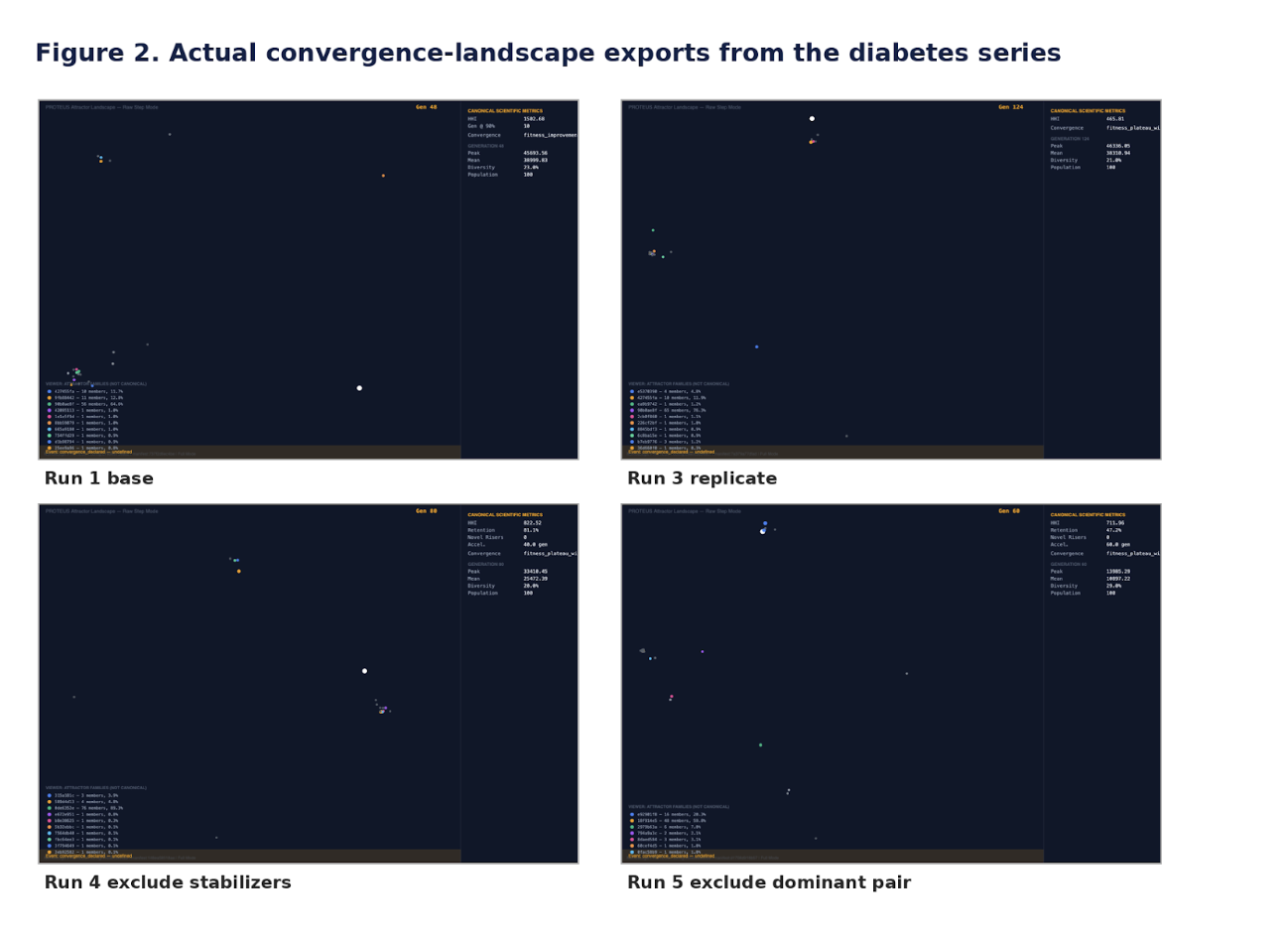
The baseline run and its same-parameter replications repeatedly returned a compact WFS1-linked family centered on rs147834269 and rs1801212, with rs1810396 and rs78142091 reappearing as recurrent support-layer variants. The importance of that result is not that these loci automatically constitute a final causal answer. The importance is that the system did not wander arbitrarily. Under identical conditions, it repeatedly found the same structural neighborhood.
That repeatability is the first important result.
In a field accustomed to unstable association patterns, repeated return to the same family matters. It suggests that the underlying search is not merely rediscovering whichever high-scoring feature happens to float to the top on a given run. It is recovering a stable interaction basin.
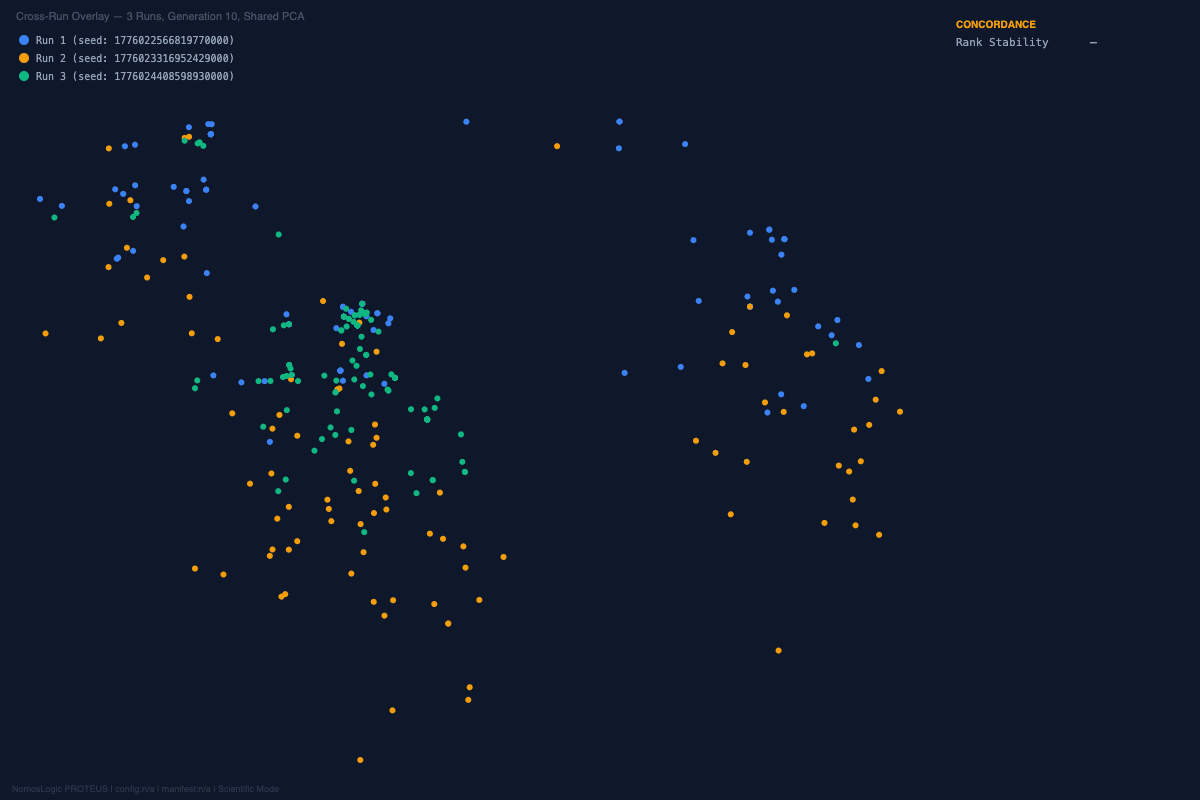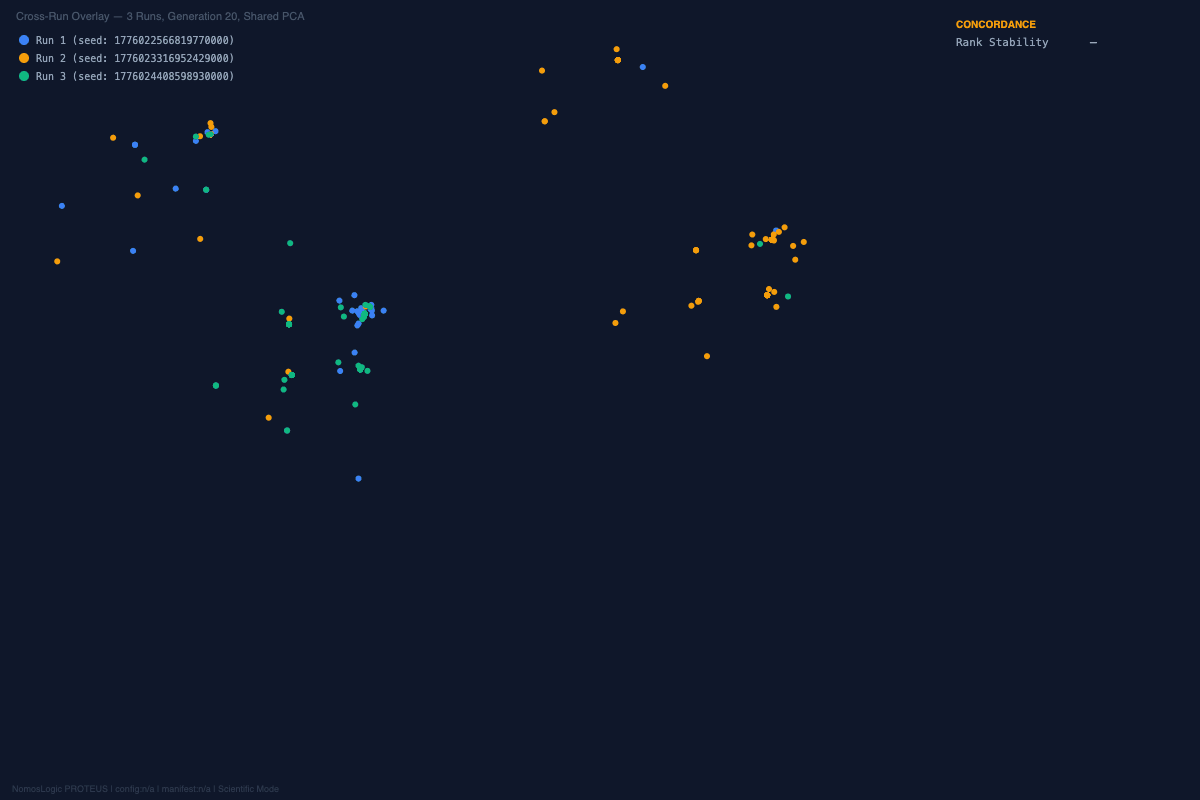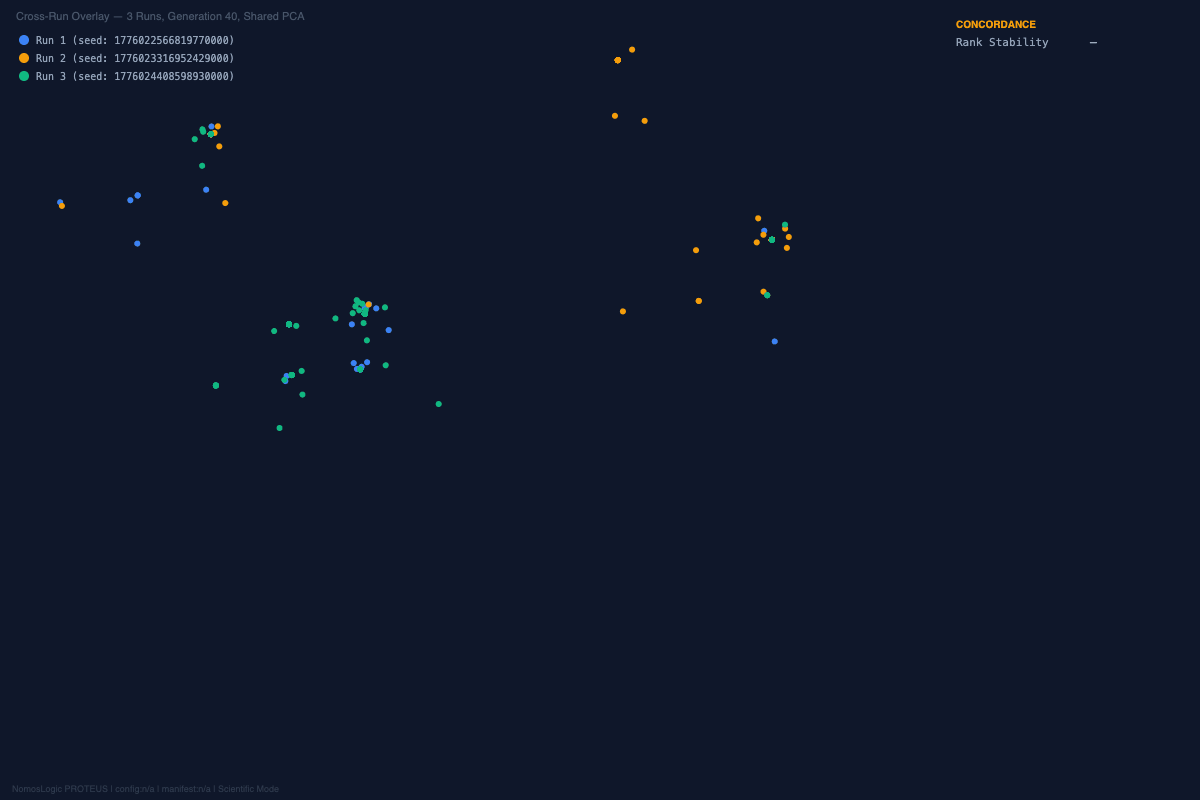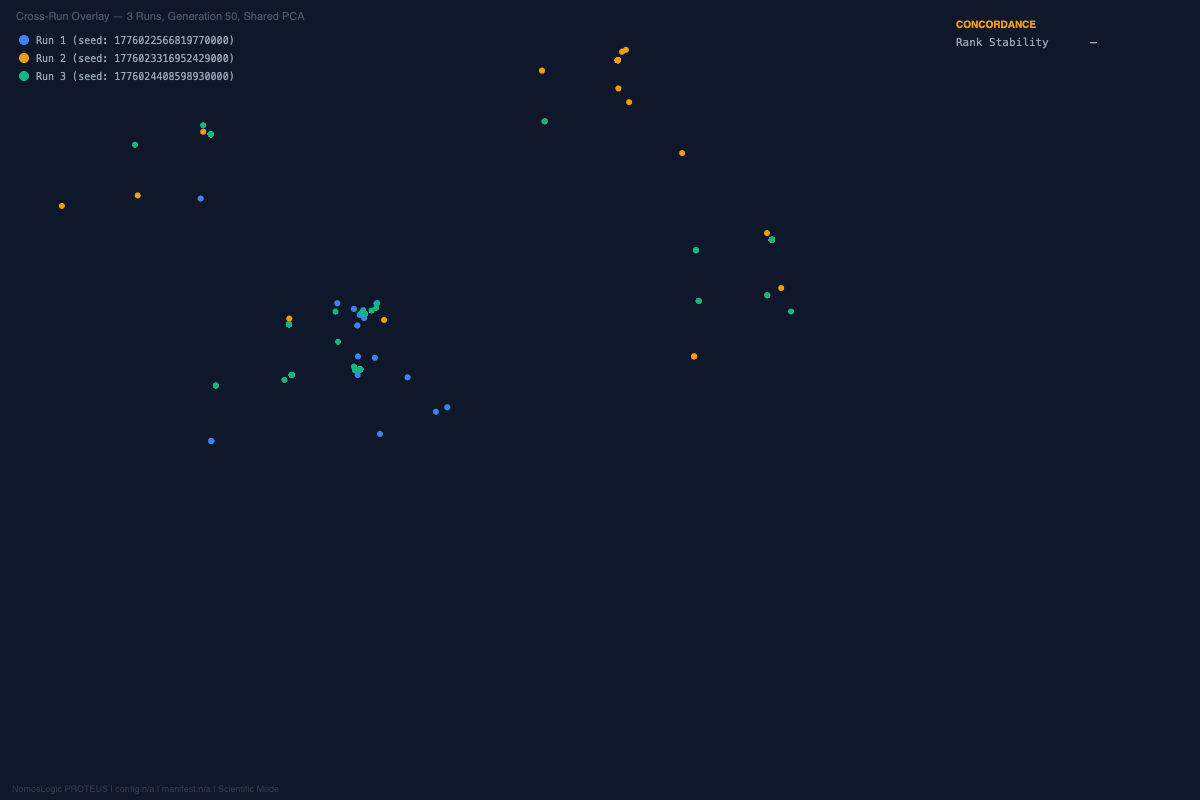
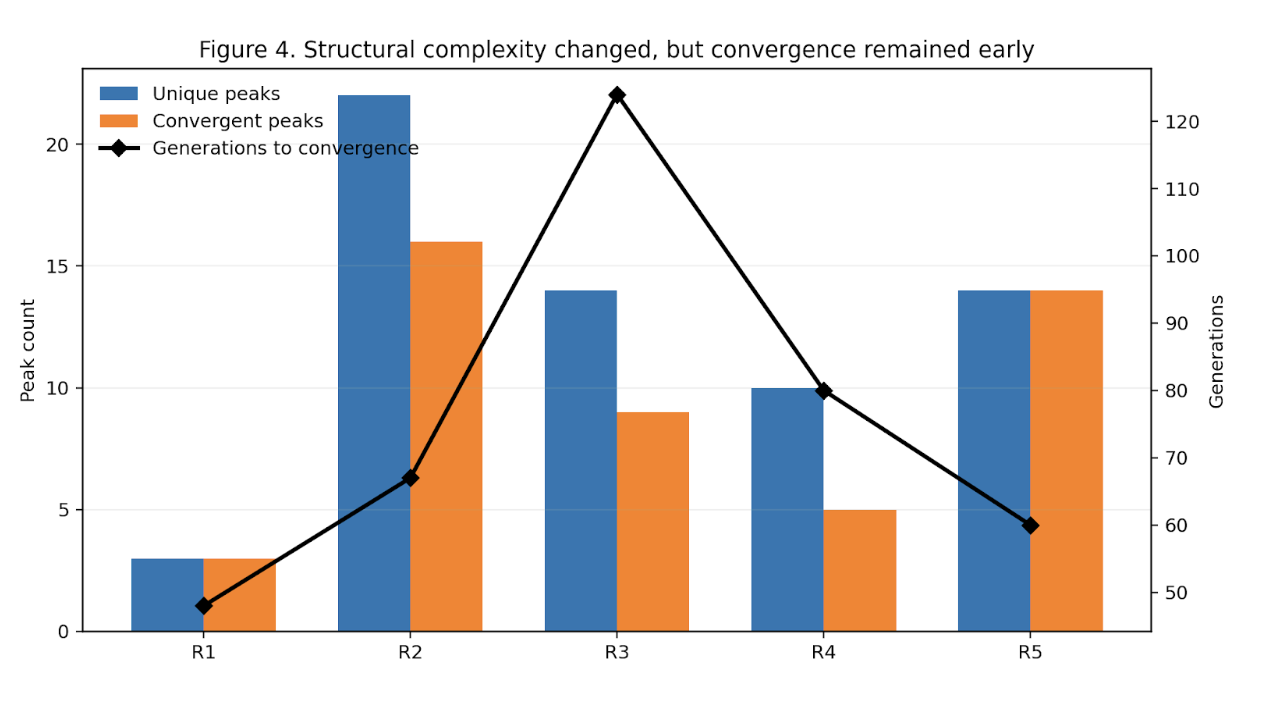
And the visual story was aligned with the structural one. Across the repeated runs, the convergence landscapes remained organized rather than diffuse. The exact peak composition shifted at the margins, but the architecture kept returning to the same core family. That is not what random optimization drift looks like. It is what constrained recurrence looks like.
The decisive test was perturbation
The stronger result came when the apparently important variants were removed.
First, the recurrent support-layer variants were excluded. The system did not fail. It simplified and reconverged, with rs1801212 remaining central and alternate companions, including rs4458523, moving upward into the new structure.
That alone was informative. It showed that the original family was not dependent on every visible stabilizer. Some contributors were recurrent, but replaceable. The architecture narrowed, but it did not collapse.
Then the more severe test was applied: exclusion of the dominant WFS1-linked pair itself.
That is the point at which a brittle model should break.
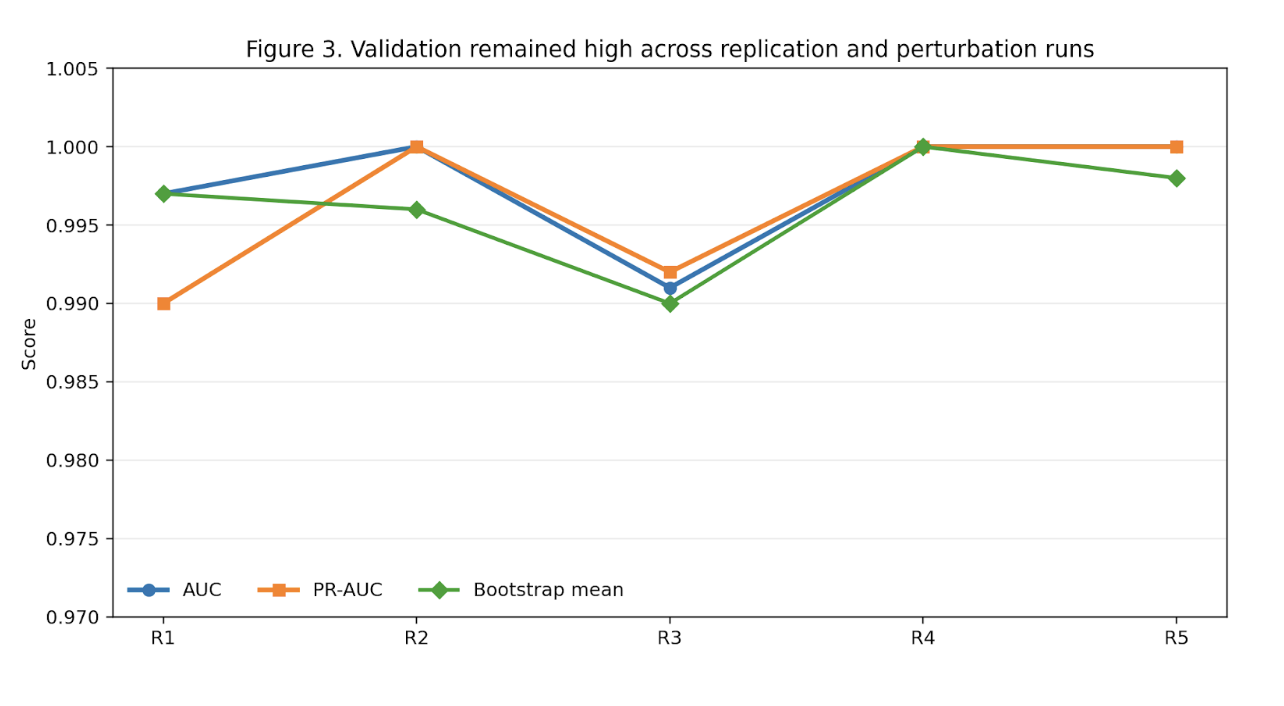
If the original architecture had been nothing more than a single fragile solution, this step should have flattened the landscape into noise, scattered the peaks into non-reproducible fragments, or forced a major loss of fitness and predictive separation. That is not what happened.
A different coherent family emerged, built around rs4458523, rs2071403, rs1810396, and often rs78142091. The top family changed. The system behavior did not.
That is the key result of the study.
The architecture was not invariant, but it was not fragile either. It reorganized into an alternative high-fitness configuration. That is exactly the kind of behavior one expects from a distributed constraint system and not from a single-driver story.
In other words, the strongest visible pair was important, but it was not uniquely indispensable.
What makes this more than a variant-ranking exercise
This is where the diabetes study stops being “another genomic result” and starts becoming methodologically important.
Traditional interpretation tends to take the highest-ranked signal and quietly elevate it into the explanation itself. The top locus becomes the mechanism. The strongest pair becomes the disease story. The most enriched feature becomes the target.
But this study argues for a different discipline.
A deterministic convergence framework does not deny complexity. It disciplines complexity. It asks whether the system repeatedly resolves into stable interaction structures, whether those structures persist under perturbation, and whether alternative optima can be recovered when the apparent core is removed.
That is a much stronger test of structure than simple rank order.
The question is not merely which variant scored highest. The question is whether the architecture survives targeted disruption, and if it does, how it reconstitutes itself. That is a far more useful way to think about biological robustness than a single p-value attached to a single marker.
The study also shows what caution looks like
This result should not be overstated.
It does not prove universal biological determinism. It does not establish strict interventionist causality from perturbation alone. It does not mean every recurrent family is automatically mechanistic in the narrowest sense. And it does not remove the need for rigorous mapping discipline at the variant level.
The persistent rs121912648 block remains a separate unresolved issue and should be treated as such rather than casually folded into the main mechanistic story. Its persistence across runs makes it structurally important, but its interpretation remains distinct from the cleaner WFS1-centered and alternate reconfigured families.
That is not a weakness in the framework. It is evidence of scientific discipline.
Because the right conclusion is not “everything is solved.” The right conclusion is narrower and stronger:
Under identical conditions, this diabetes system repeatedly converged on stable families; under targeted perturbation, it reorganized into alternative coherent families rather than collapsing.
That is already a serious claim.
Why this matters for drug discovery
The importance of this result is not confined to diabetes.
It speaks to a broader methodological problem in genomics and drug discovery. We keep elevating the strongest visible signal into the explanation itself. Then, when that signal fails to replicate cleanly or fails to translate into intervention, we conclude that biology is noisy.
Often the better conclusion is that biology is structured differently than our models assume.
If a genomic system repeatedly reconstructs itself after apparent core disruption, then a single-target framework is not describing the architecture well enough. It may still identify something biologically real, but it will not tell us whether that signal is an anchor, a bridge, a support layer, or a replaceable expression of a deeper constraint structure.
That distinction is not academic. It is exactly the distinction that matters when programs fail because the “best target” turns out not to be structurally necessary.
A deterministic convergence framework changes the objective. Instead of asking only what is enriched, it asks what is recurrent, what is structurally central, what is replaceable, and what genuinely breaks the system when removed.
That is a more useful discovery logic.
It favors robust targets over fragile associations. It makes compensatory pathways visible before they sabotage development. And it pushes the field away from static correlation lists and toward observed system behavior.
What the field should take from this diabetes series
The lesson is not that WFS1 is the whole story.
The lesson is that the story is architectural.
We should be looking for recurrent families, bridge variants, support layers, compensatory structures, and points of actual structural failure. We should stop treating the top-ranked contributor as if it were automatically the mechanism. We should stop assuming that the strongest visible signal is the same thing as the irreducible biological core.
That is why this diabetes study matters.
It did not simply produce another set of markers. It forced a harder and better question:
When you remove the signal you think matters most, what does the system do next?
In this case, the answer was not collapse.
It was reorganization.
And that may be the more important biological fact.