"Probability tells you what to believe. Mechanism tells you what to do."
That is the epigraph of my new book. It is also the line that separates the next decade of genomics from the last twenty years.
Deterministic Convergence: Biological Systems, Architecture, and the Search for Hidden Order is live on Kindle today. Paperback and hardcover are shipping soon.
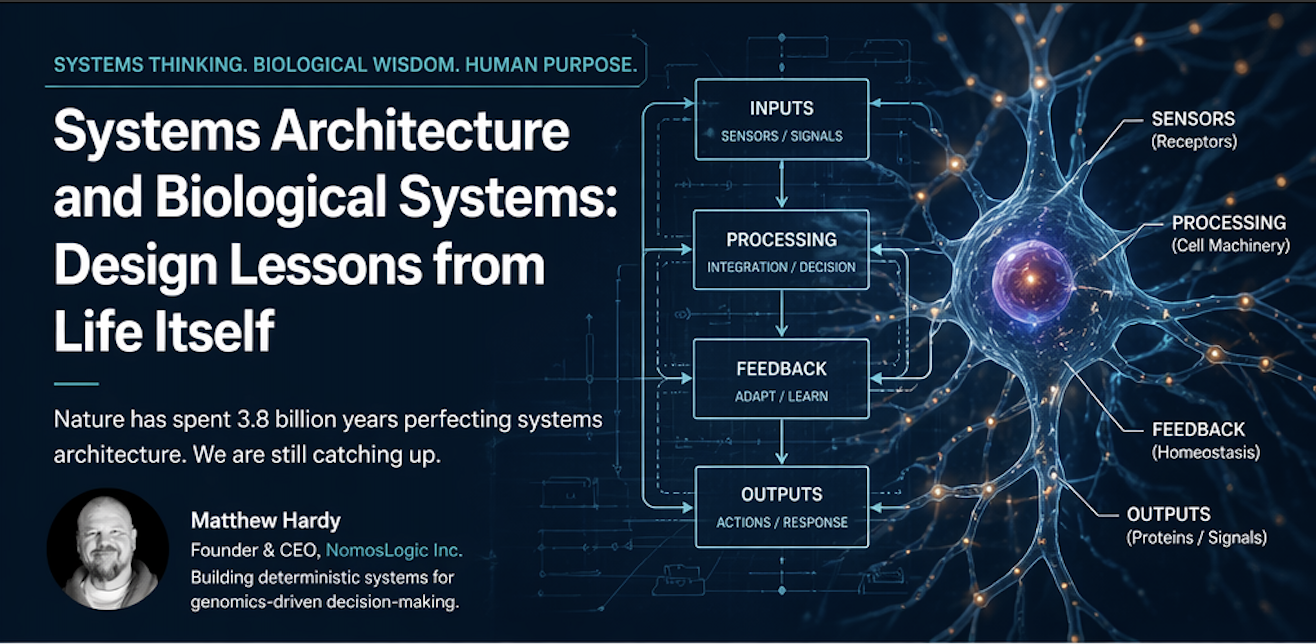
The argument is structural. Modern genomics has built an enormous machinery for producing parts and a much smaller machinery for understanding what those parts mean when placed back inside a system. That is the fragmentation error: treating parts as explanations, lists as architectures, associations as mechanisms, probabilities as causes. The cost shows up in three places that matter operationally: the clinic, drug discovery, and population health.
The book introduces two operationalized concepts, deterministic convergence and distributed constraint architecture, validated across six clinical domains: cardiovascular, neurological, oncological, renal, metabolic, and hematological. Same analytic operation, six different systems, consistent architectural behavior in every case.
This is not a manifesto. It is a foundational reference. Methodology layer, worked examples, formal definitions, explicit failure modes, comparisons to adjacent frameworks, and a research program for the work that remains.
Architecture, not inventory. Mechanism, not pattern. Determinism, not probability.
The running is for others to do.
Excerpt:
"Patient stratification is one of the most direct clinical applications of
architectural analysis. Patients with the same phenotypic diagnosis
frequently have different underlying architectures. These architectural
differences produce different responses to intervention, different disease
trajectories, and different prognostic profiles. Fragment-centered analysis
has limited ability to resolve these differences. Architectural analysis can
make them visible and actionable.
The clinical challenge of phenotypic heterogeneity is well-recognized.
Oncology has organized around this challenge for decades, subdividing
what used to be called cancer into increasingly specific molecular and
architectural categories. Cardiovascular medicine has recognized for years
that patients with similar risk scores can have very different underlying
biology. Psychiatry has struggled with the heterogeneity within diagnostic
categories like major depression or schizophrenia. Metabolic medicine has
the same challenge with type 2 diabetes, which clearly includes multiple
distinct pathophysiologies under a single label.
In each of these domains, the current stratification approaches use some
combination of molecular markers, clinical features, treatment response
patterns, and emerging omics data. The approaches have produced
meaningful advances. They have also, consistently, left substantial
unexplained variability. The residual variability suggests that the
stratification dimensions currently in use do not capture the full
architectural reality."