Hematology Study Commentary Nested distributed constraint architecture under progressive multi-locus exclusion
NOMOSLOGIC STUDY COMMENTARY
NomosLogic | Proteus Commentary | April 2026
The hematology study reveals a system that behaves less like a checklist of isolated variants and more like an engineered architecture with layered reserve capacity. Under baseline conditions, the model resolves into a high-fitness, strongly validated hematologic configuration. As dominant contributors are progressively removed, the system does not immediately fail. Instead, it compresses through smaller fallback basins, exposing hidden compensatory structure. Only after exclusion of multiple primary and reserve contributors does the architecture fall out of its earlier high-fidelity regime, with sharp degradation in peak fitness, discrimination, calibration, and stability. This is the central result: hematologic behavior in this cohort appears governed by nested distributed constraints rather than simple single-locus dependency. In practical terms, the study suggests that true hematologic risk and resilience may be encoded in interacting systems, not isolated flags, and that meaningful breakdown only emerges once enough of the compensatory architecture is removed.
Executive thesis. This study demonstrates that the hematologic system does not behave like a brittle single-locus model. Instead, it exhibits layered compensatory behavior: early perturbations reduce fitness while preserving convergence, later exclusions compress the architecture into smaller fallback basins, and only after removing multiple primary and reserve contributors does the system fall out of its earlier high-fidelity regime. The result is strong evidence for nested distributed constraint architecture rather than simple variant dependence.
Study summary
Study objective. To determine whether the hematology system is dominated by one or two primary contributors, or whether it contains layered reserve structure capable of preserving convergence under progressive exclusion of high-weight loci.
Method. The study began with a stable baseline hematology architecture and then applied successive multi-locus exclusions. Each run was assessed for peak fitness, convergence timing, recurrent peak structure, ROC and PR performance, calibration behavior, and bootstrap stability.
Headline finding. The system repeatedly reorganized into smaller and weaker but still coherent basins until a seven-locus exclusion finally produced a clear structural break. That progression is more consistent with a finite layered system than with a simple single-path model.
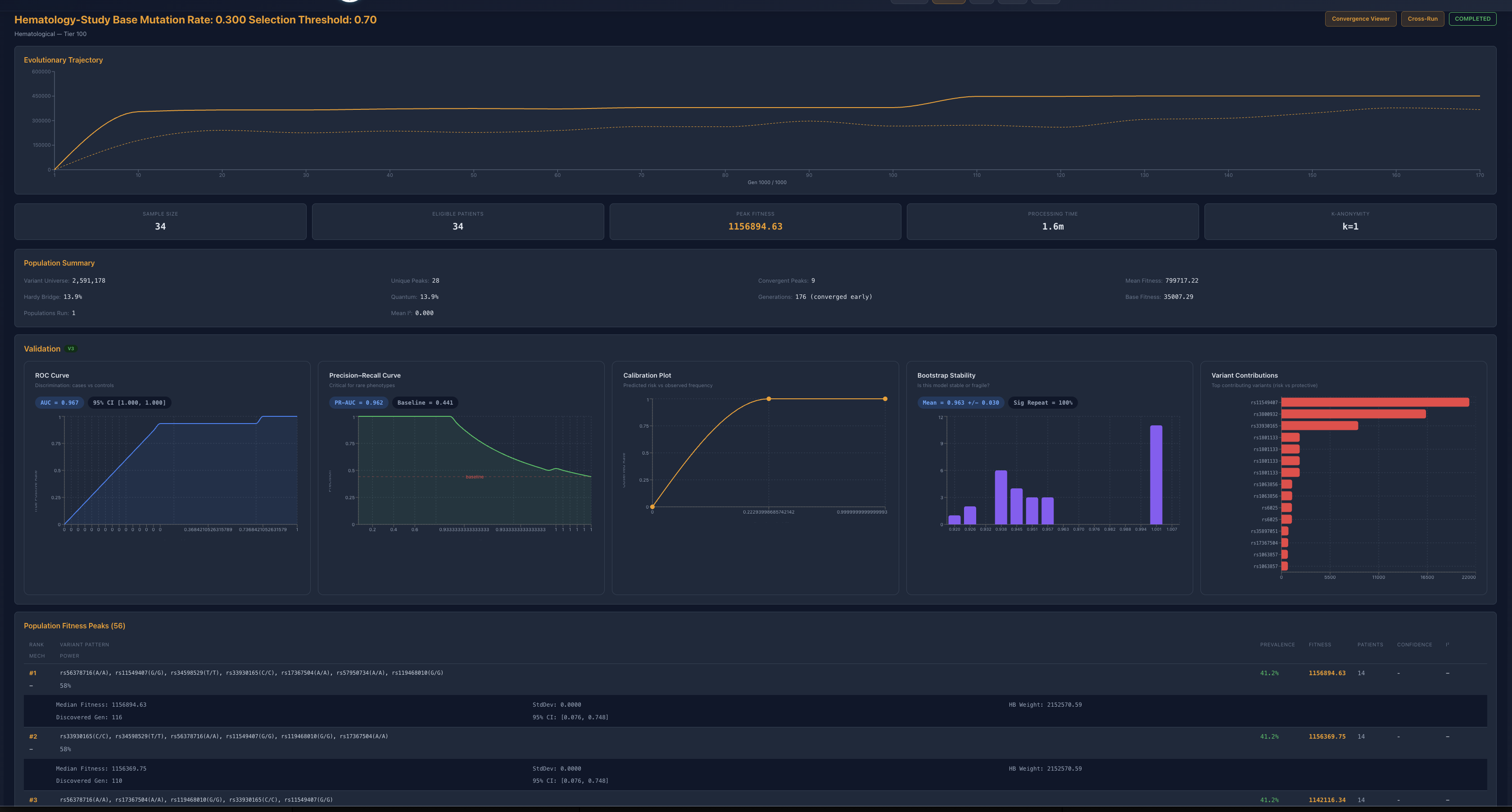
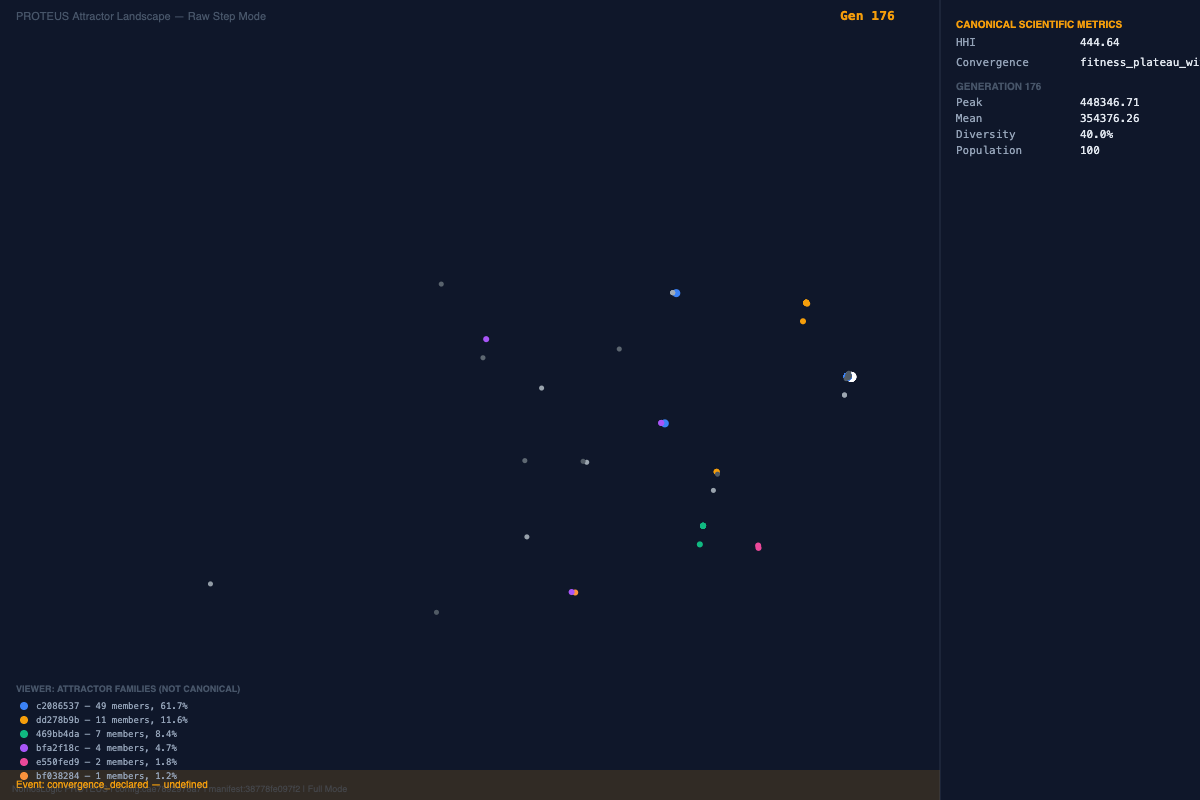
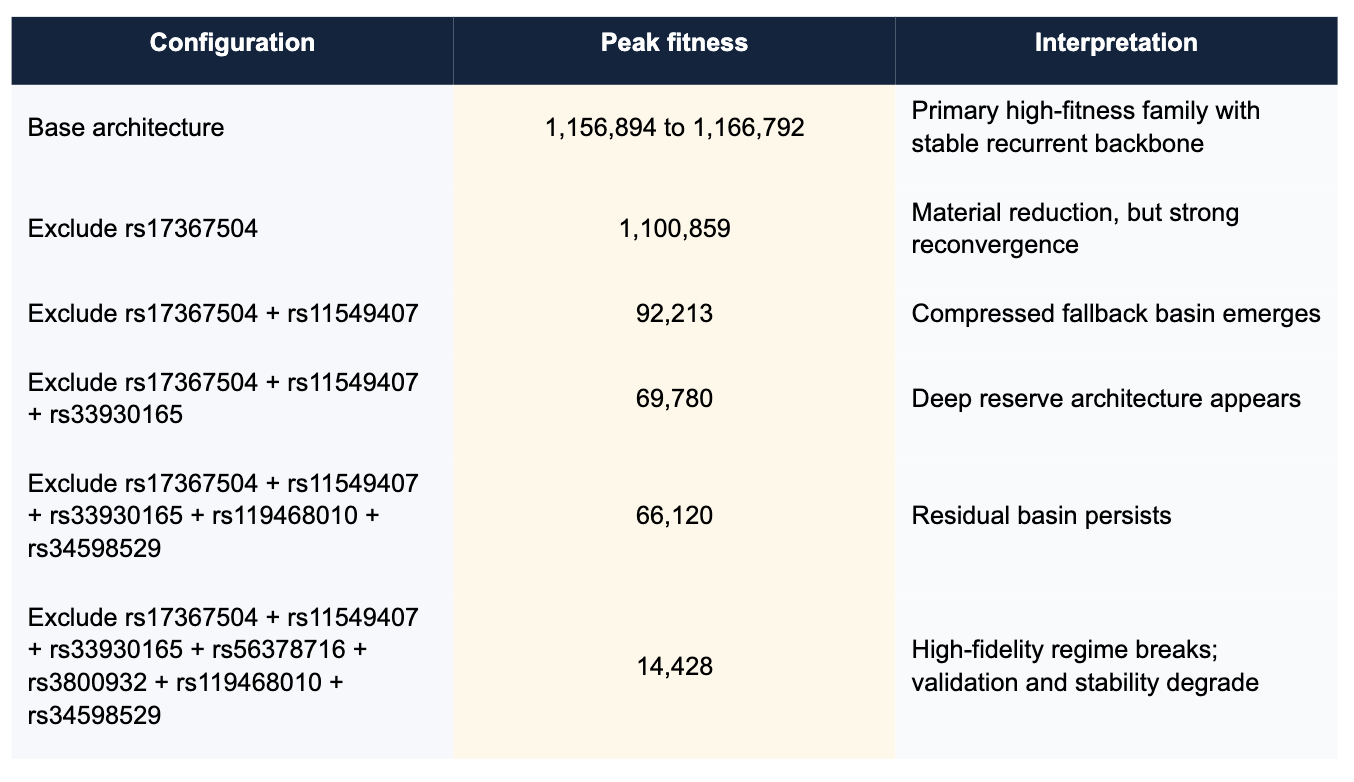
Figure 1. Baseline hematology architecture. The base run resolves into a high-fitness family with strong validation and a stable recurrent contributor backbone.
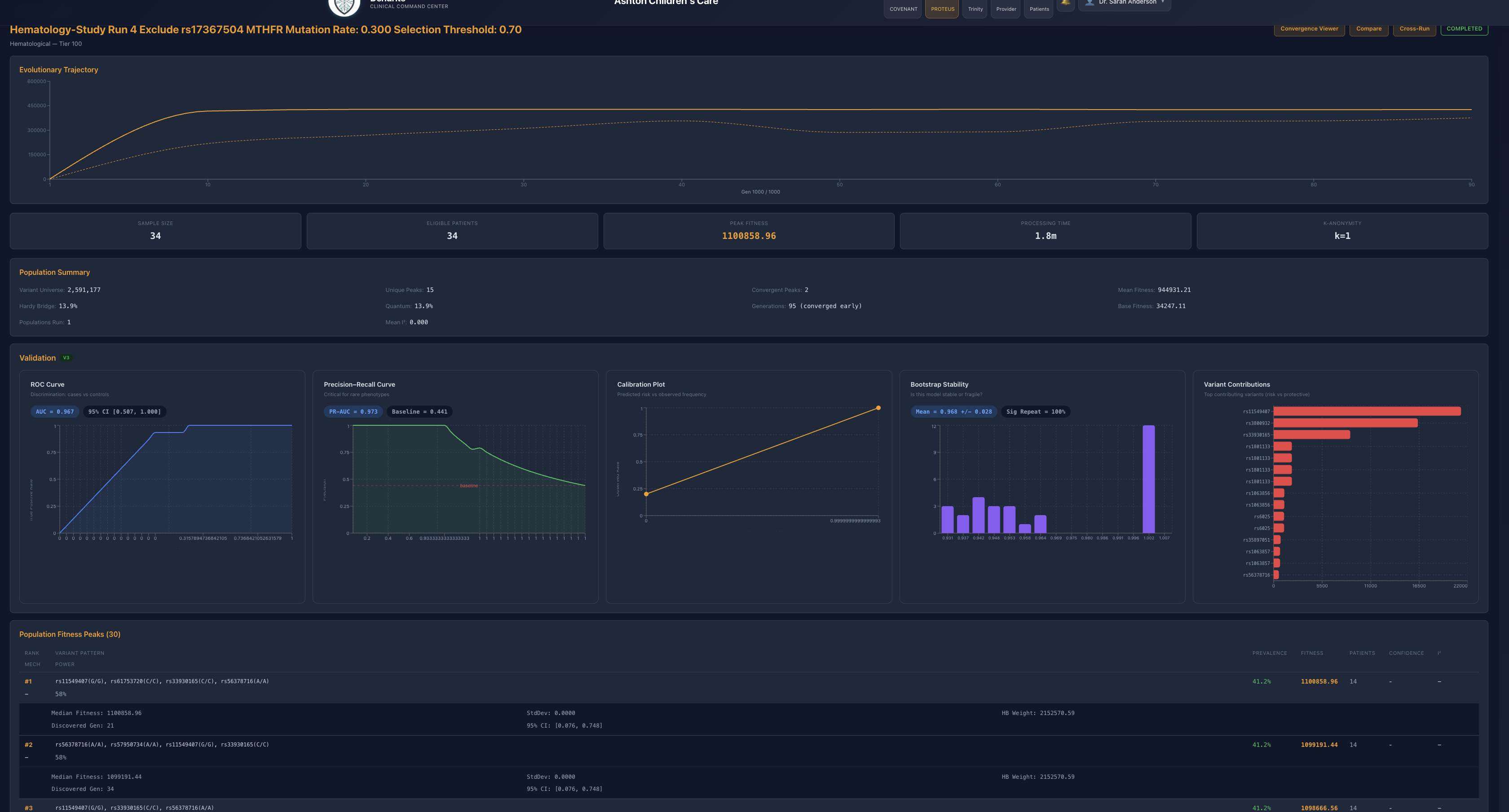
Figure 2. Single exclusion of rs17367504 (MTHFR). Fitness falls, but the system remains highly convergent, indicating that rs17367504 is contributory rather than singularly decisive.
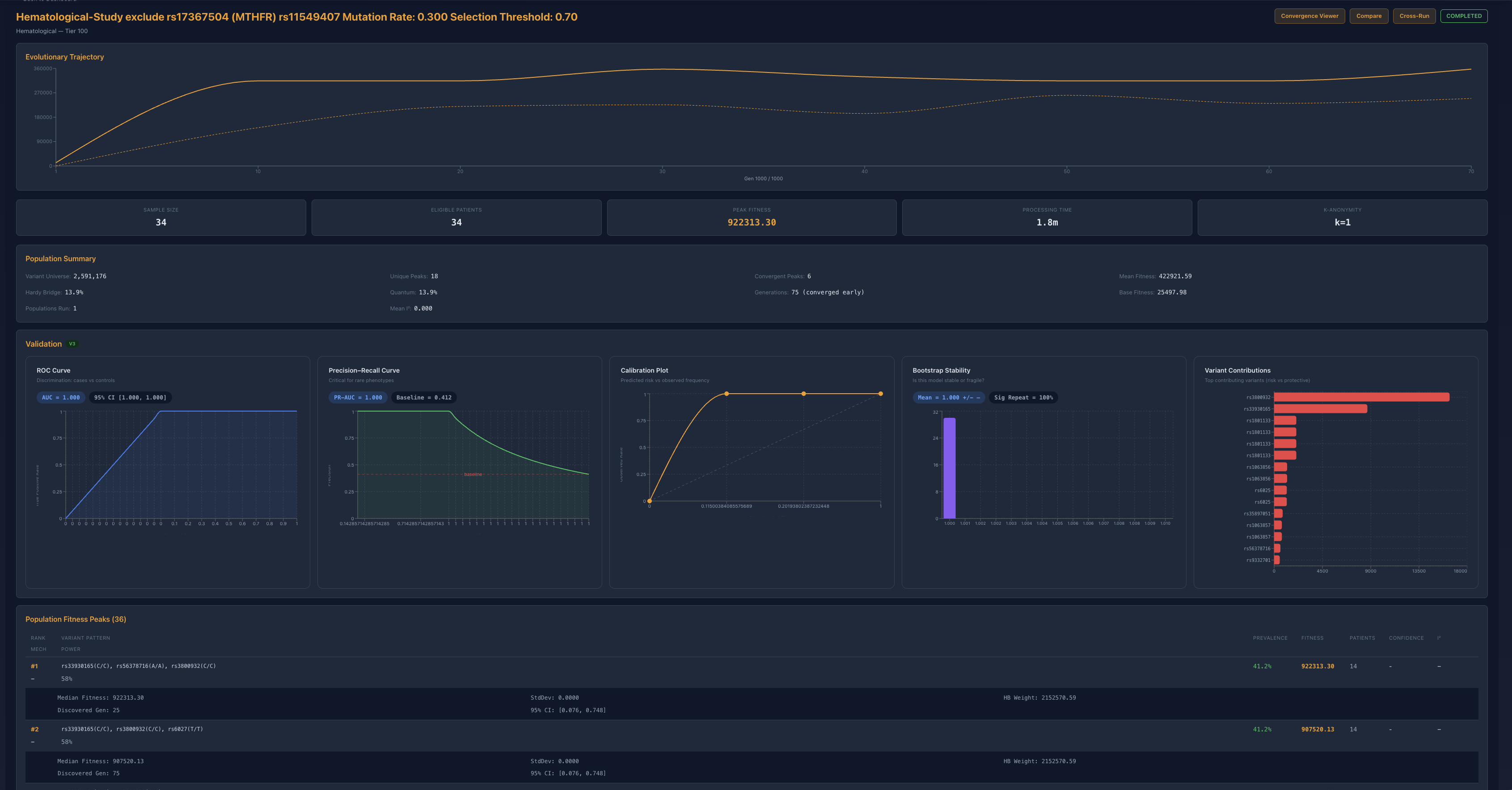
Figure 3. Dual exclusion of rs17367504 and rs11549407. The architecture compresses into a fallback basin rather than collapsing, exposing reserve structure.
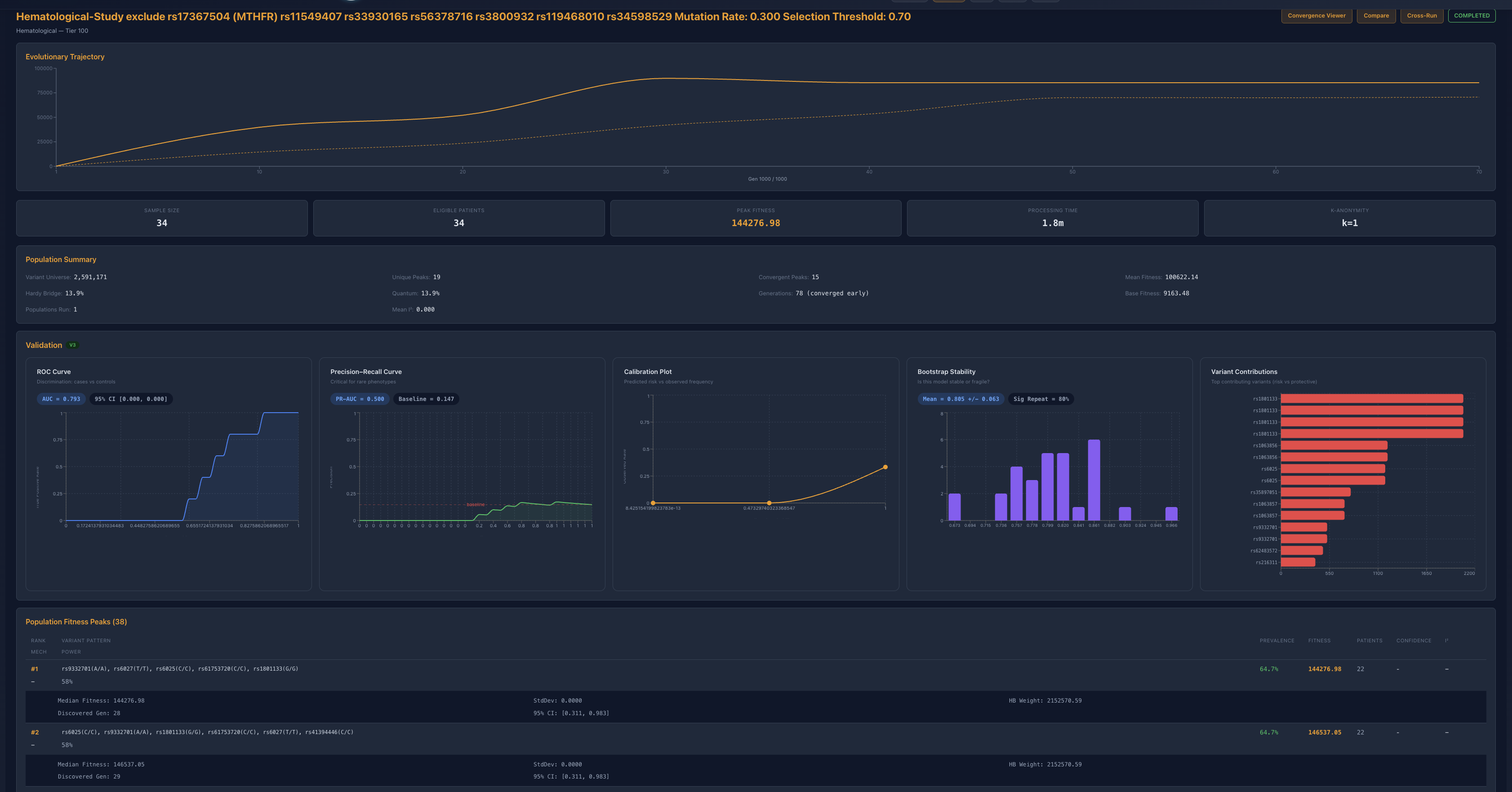
Figure 4. Seven-locus exclusion. Peak fitness, discrimination, calibration, and stability all degrade, marking the first clear break from the earlier high-fidelity regime.
In-depth commentary
Baseline behavior
The baseline hematology study resolved to a high-fitness family around 1.16 million fitness units, with near-perfect ROC and PR performance and tightly repeating dominant peaks. This establishes a legitimate stable regime rather than a fragile one-off solution. Importantly, repeated base runs preserved a shared structural backbone while allowing modest peripheral variation, which is already suggestive of constrained reconfiguration rather than random drift.
Single-locus perturbation
Excluding rs17367504, an MTHFR-associated blood-pressure-linked contributor, reduced peak fitness but did not materially harm convergence or validation. The system quickly reassembled around a still-strong architecture. This is the first sign that rs17367504 is meaningful but not singularly load-bearing.
Dual exclusion and architectural compression
When rs11549407 was removed alongside rs17367504, the system took a much larger fitness hit and compressed into a smaller fallback set. The key point is that the study still reconverged cleanly. A brittle single-locus interpretation should have failed here. Instead, the system rerouted into a lower-power but structurally coherent basin.
Triple exclusion and deep reserve architecture
Excluding rs33930165 in addition to the prior pair pushed the system deeper into reserve behavior. Peak fitness fell again and convergence took longer, but the architecture still found a recurrent solution. This establishes that the hematology system contains not merely one fallback layer, but nested compensatory structure.
Residual basin under reserve-pair attack
Removing rs119468010 and rs34598529 after the earlier exclusions only modestly reduced peak fitness further. That is a crucial result: these reserve contributors mattered, but they were still not the last irreducible support. At this stage the system had shed multiple primary and secondary contributors and was still able to reconverge.
Collapse boundary
The decisive step came when rs56378716 and rs3800932 were also removed alongside the prior exclusions. Peak fitness collapsed to roughly 14.4k, ROC fell to 0.793, PR-AUC degraded to 0.500, bootstrap stability broadened to 0.805 +/- 0.063, and significant-repeat behavior fell to 80 percent. The algorithm still found a mathematical basin, but it was no longer the same class of stable, high-fidelity, biologically legible architecture seen earlier. This is the first clear transition out of the prior regime.
Interpretive conclusion
Taken together, the perturbation ladder supports a finite but deeply layered hematologic system. The architecture is neither single-point fragile nor infinitely compensatory. It maintains function across several rounds of exclusion by compressing through primary, fallback, reserve, and residual structures, and then eventually loses coherent high-quality resolution once enough load-bearing contributors are removed. That pattern is the signature of nested distributed constraints.
Why this matters
• It argues against simplistic single-variant causality in hematologic system behavior.
• It shows that clinically relevant architecture can remain stable despite removal of apparently dominant contributors.
• It identifies a measurable collapse boundary, which is much more informative than simple survival under one perturbation.
• It gives Proteus a stronger systems-level story: not just resilience, but hierarchical resilience with finite limits.
• It creates a cleaner bridge to drug discovery and precision medicine by showing which contributors are dominant, which are compensatory, and which combinations finally break the regime.